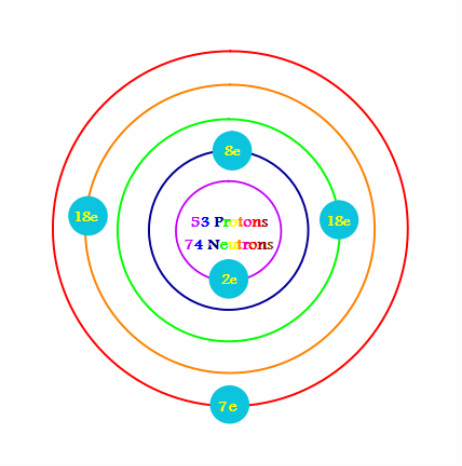
Bohr Diagram of Iodine
How do you create a Bohr Diagram? Well, first you must know how many protons, electrons, and neutrons the element has. The way you are able to figure out that piece of information is to first locate the atomic number and atomic mass. By just looking at the atomic number, right off the bat we know that the number of protons is the same as the atomic number. For example, in the element Iodine, the atomic number is 53, so that way we know that there are 53 protons. Then, we could assume that the number of the protons and the numbers of the electrons are equivalent. With that being said, we also know that there are 53 electrons. Next, you round the atomic mass to the nearest whole number and then subtract the atomic mass from the atomic number. The difference will determine how many neutrons the element has. Now, you have to know the maximum amount of electron each electron cloud holds. (Two in the first, eight in the second, eighteen in the third, thirty-two in the fourth, fifty in the fifth, and so on.) Then draw your nucleas in the center and write in how many protons and neutrons there are. Next, decide how many electron clouds you'll need, draw them, and write how many electrons would be in each cloud. Now, you officially created a Bohr Diagram!